Several BCRF researchers participate in early conference sessions discussing the latest in their research
Greetings from SABCS!
The San Antonio Breast Cancer Symposium (SABCS) kicked off its 39th annual meeting this week. The BCRF research program and communications team are attending the meeting, along with thousands of physicians, researchers, health care professionals and advocates, to hear the latest in breast cancer research and news. We will be posting daily blogs as well as lighting up social media with highlights from the entire week. We hope you will follow along!
The meeting started with education sessions on Tuesday, prior to the official start Wednesday morning. A primer to the meeting itself, the education sessions were presented by experts on key issues and updates in breast cancer research and patient care. BCRF investigators Drs. Powell Brown, Jenny Chang, Arti Hurria, Melinda Irwin, Seema Khan, Peter Lee, Ann Partridge, Charles Perou, Kornelia Polyak and Rachel Schiff spoke in their respective fields of research, many highlighting their BCRF-supported work.
Wednesday Highlights – Updates in management of metastatic ER-positive breast cancer
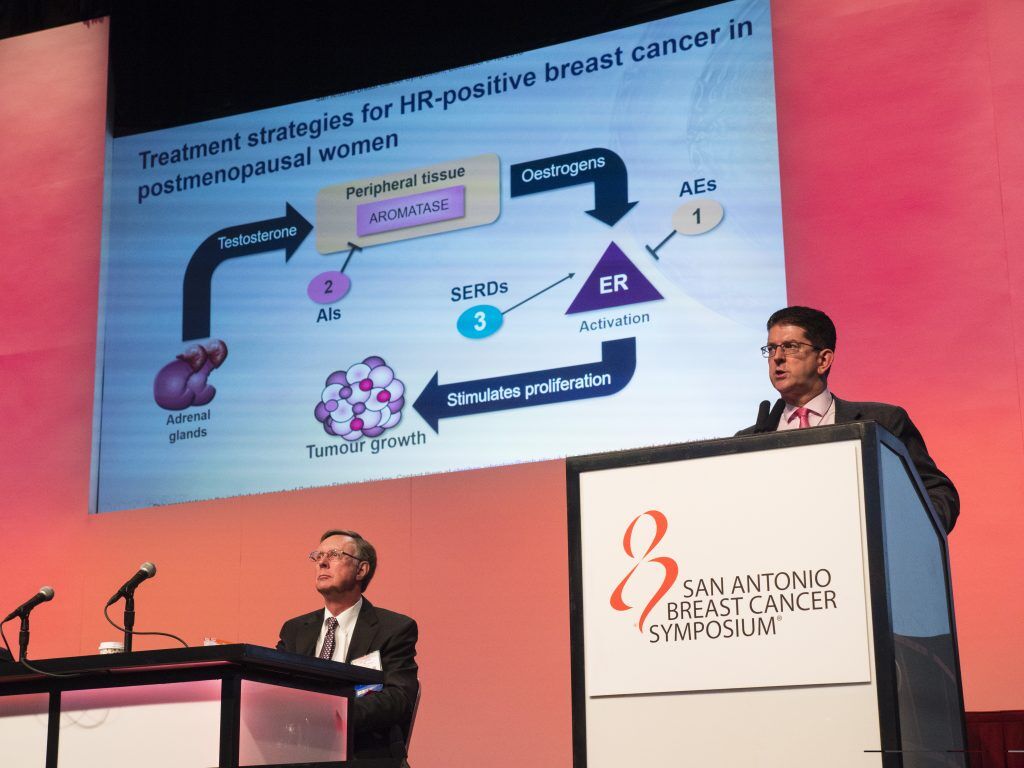
Metastatic ER+ breast cancer remains the most common form of metastatic breast cancer (MBC). Patient outcomes in ER+ MBC depend on a variety of clinical factors. One of the prevailing challenges has been tumor resistance to anti-estrogen therapies, tamoxifen and aromatase inhibitors (AI). In the opening plenary session, which set the stage for a larger discussion on the management of ER+ MBC, Dr. Stephen Johnston of the Royal Marsden Hospital, UK discussed how to optimize hormone treatment in ER+ MBC, focusing on new combination therapies, as well as laboratory research that has identified tumor mutations responsible for resistance to AI. Recent trials (PALOMA 3 and MONALEESA), which combine an AI with a CDK4/6 inhibitor, have demonstrated a clear benefit in ER+ MBC, but questions remain as to whether every patient requires the combination. Mutations in ESR1, the gene that codes for the estrogen receptor, and PIK3CA following treatment with an AI has been shown to induce resistance to AI therapy. While studies continue to identify potential drivers of resistance that may be targetable, Dr. Johnston cautioned that the clinical challenge will be in understanding how best to apply the new knowledge and combination approaches in devising the most appropriate therapy for each patient.
Dr. Ofir Cohen of the Dana Farber Cancer Institute immediately followed Dr. Johnston’s talk with a presentation on new data showing that drug-resistant ER+ MBC tumors have a unique genetic profile from the initial (primary) tumor. In work supported in part by BCRF, the Dana Farber team, including BCRF investigator Dr. Nikhil Wagle, performed genetic sequencing on metastatic tumors from 88 patients, 27 for whom they had matching DNA from the primary tumor. The analysis identified mutations in several important tumor-associated genes (ESR1, ERBB2 (HER2), PIK3CA, PTEN, RB1 and AKT) that were increased in the metastatic tumors compared to tumors htat had not been previously treated. Clinically relevant results of the genetic studies are being returned to the patient and clinician to inform clinical decision making. In a press release about the study, Dr. Wagle commented: “The goal is to integrate the functional and clinical findings into a unified ‘Resistance Atlas’ for ER-positive MBC, which should help inform treatment decisions for individual patients as well as propel the development of new treatment strategies for ER+ MBC.”
In trial results from the ECOG group (PrECOG 0102), including BCRF investigators Drs. Kathy Miller, Joseph Sparano and Antonio Wolff, post-menopausal women with ER+MBC receiving the mTOR inhibitor everolimus with the AI fulvestrant had a two-fold increase in progression free survival — a measurement of time without progression of disease — compared to women who did not receive the mTOR inhibitor. All women on the study had previously progressed on AI therapy alone. Commenting on the study for an SABCS press release, study author Dr. Noah Kornblum of Montefiore-Einstein Center for Cancer Care, NY cautioned: “We have to resist temptation to immediately adopt a positive result of a novel combination from a small study into a new standard of care. It may be one day the case that everolimus and fulvestrant becomes a new approved therapy for ER+ MBC, but caution is prudent until larger studies confirm these results.” The authors noted that grade 3/4 side effects related to everolimus were reported in 53 percent of patients receiving the mTOR inhibitor and that patients and clinicians should be aware of potential complications with the addition of a mTOR inhibitor to standard care.
Reporting for the NSABP trial group (NRG Oncology), Dr. Terry Mamounas of the University of Florida Cancer Center, Orlando, FL presented data from the NSABP B-42 trial showing that five additional years of the AI letrozole did not provide significant benefit in overall survival in women with early-stage ER+ breast cancer with five years of previous endocrine therapy (tamoxifen or AI). There was, however, a significant decrease in the risk of breast cancer recurrence or cancer in the opposite breast with the additional five years of letrozole. The study involved nearly 4,000 patients with seven years of follow up.
Continue to follow BCRF at SABCS on our social media channels (Facebook, Twitter and Instagram)! Check back tomorrow for the latest highlights of #SABCS16.